The Effect of Macronutrient Composition on Endogenous Long-Chain Polyunsaturated Fatty Acid Status during Weight Loss
This article at a glance  The global obesity epidemic is still getting worse in most parts of the world, as highlighted in recent reports. Addressing the obesity problem is important because being overweight is associated with increased mortality from a range of metabolic disorders, but on the other hand it potentially can be addressed. Obesity-associated disease is not uniform across populations, stimulating interest in understanding how weight gain prevention and promotion of weight loss can be best addressed. An unresolved issue is whether chronic excessive intake of easily available carbohydrates promotes obesity prevalence. Similarly, it is important to know if lowering the intake of carbohydrates in overweight individuals can contribute to weight loss. Further, with carbohydrates being important macronutrients, would lowering their intake in overweight people have any unintended effects? Hyperglycemia and hyperinsulinemia following chronically-elevated dietary intake of carbohydrates are responsible for increased fat storage and insulin resistance due to accelerated oxidation of circulating glucose to reactive advanced glycation end-products in vascular tissues. The inflammation of metabolically-active tissues has also been implicated in dysregulated metabolic responses to elevated carbohydrate intake. In contrast with these hypotheses, however, are views that indicate that these mechanisms are not of sufficient magnitude or duration to modify the processes the body uses to handle excess energy intake or sugars specifically. Furthermore, much of the evidence linking carbohydrate intake and weight gain is based on observational and cross-sectional studies, whereas intervention studies that could provide a more definite answer have been small in sample size, of too short duration, or did not control for potential confounders. Although intuitively a simple relationship, at present it is still a leap to conclude that a diet with a low glycemic load can prevent weight gain or significantly contribute to weight loss. Several methodological limitations need to be overcome to allow drawing solid conclusions on the relationship between carbohydrate intake, glycemic index and load, and weight gain or loss. For a start, prospective studies need to adequately monitor food intake. Food records are preferred over the use of food-frequency questionnaires that rely on people’s (generally poor) memories of past dietary habits, and cognitive ability. Food records also allow a better recording of the specific details on how food was prepared, which affects the glycemic index and load of food items rich in carbohydrates. However, maintaining a food record by study participants carries a risk that dietary habits may change, generally towards currently fashionable dietary recommendations. Furthermore, measurement error should be eliminated through professional supervision guiding participants on how to fill out their records. Ideally, multiple dietary records should be taken that span a sufficiently long period and cover the different seasons in studies lasting more than a year.
The global obesity epidemic is still getting worse in most parts of the world, as highlighted in recent reports. Addressing the obesity problem is important because being overweight is associated with increased mortality from a range of metabolic disorders, but on the other hand it potentially can be addressed. Obesity-associated disease is not uniform across populations, stimulating interest in understanding how weight gain prevention and promotion of weight loss can be best addressed. An unresolved issue is whether chronic excessive intake of easily available carbohydrates promotes obesity prevalence. Similarly, it is important to know if lowering the intake of carbohydrates in overweight individuals can contribute to weight loss. Further, with carbohydrates being important macronutrients, would lowering their intake in overweight people have any unintended effects? Hyperglycemia and hyperinsulinemia following chronically-elevated dietary intake of carbohydrates are responsible for increased fat storage and insulin resistance due to accelerated oxidation of circulating glucose to reactive advanced glycation end-products in vascular tissues. The inflammation of metabolically-active tissues has also been implicated in dysregulated metabolic responses to elevated carbohydrate intake. In contrast with these hypotheses, however, are views that indicate that these mechanisms are not of sufficient magnitude or duration to modify the processes the body uses to handle excess energy intake or sugars specifically. Furthermore, much of the evidence linking carbohydrate intake and weight gain is based on observational and cross-sectional studies, whereas intervention studies that could provide a more definite answer have been small in sample size, of too short duration, or did not control for potential confounders. Although intuitively a simple relationship, at present it is still a leap to conclude that a diet with a low glycemic load can prevent weight gain or significantly contribute to weight loss. Several methodological limitations need to be overcome to allow drawing solid conclusions on the relationship between carbohydrate intake, glycemic index and load, and weight gain or loss. For a start, prospective studies need to adequately monitor food intake. Food records are preferred over the use of food-frequency questionnaires that rely on people’s (generally poor) memories of past dietary habits, and cognitive ability. Food records also allow a better recording of the specific details on how food was prepared, which affects the glycemic index and load of food items rich in carbohydrates. However, maintaining a food record by study participants carries a risk that dietary habits may change, generally towards currently fashionable dietary recommendations. Furthermore, measurement error should be eliminated through professional supervision guiding participants on how to fill out their records. Ideally, multiple dietary records should be taken that span a sufficiently long period and cover the different seasons in studies lasting more than a year.  A well-recognized limitation of observational studies in evaluating the association of individual food nutrients with outcomes is the need to control for many other variables that co-vary with the nutrient of interest. However, randomized controlled studies may also suffer from this limitation if the assigned diets vary in dietary components that are not being controlled. For example, a diet aiming to evaluate low-glycemic index carbohydrates may have exchanged one food component with another containing less fiber or with a different amount of protein. All macronutrient classes, as well as micronutrients, should ideally be controlled in order to draw meaningful conclusions. Observational studies are generally limited to establishing associations. These are not causal to the endpoint, and sometimes reversed causation is at play where the outcome affects the intake of the nutrient being investigated. For example, in an observational trial that lasted 6.5 years, an increase in pancreatic cancer risk associated with high available carbohydrate and low fat intake was thought to reflect the dietary changes that are associated with subclinical disease. Controlled intervention studies are preferred when intending to dissect the relationship between carbohydrate intake and body weight changes, but these need to be of sufficient duration. Carrying out a study with sufficient study duration, incorporating the necessary measures to reduce confounding factors while maintaining participant compliance, however, constitutes a significantly greater logistical endeavour than the mostly prospective observational trials and cross-sectional studies that have been employed to date.
A well-recognized limitation of observational studies in evaluating the association of individual food nutrients with outcomes is the need to control for many other variables that co-vary with the nutrient of interest. However, randomized controlled studies may also suffer from this limitation if the assigned diets vary in dietary components that are not being controlled. For example, a diet aiming to evaluate low-glycemic index carbohydrates may have exchanged one food component with another containing less fiber or with a different amount of protein. All macronutrient classes, as well as micronutrients, should ideally be controlled in order to draw meaningful conclusions. Observational studies are generally limited to establishing associations. These are not causal to the endpoint, and sometimes reversed causation is at play where the outcome affects the intake of the nutrient being investigated. For example, in an observational trial that lasted 6.5 years, an increase in pancreatic cancer risk associated with high available carbohydrate and low fat intake was thought to reflect the dietary changes that are associated with subclinical disease. Controlled intervention studies are preferred when intending to dissect the relationship between carbohydrate intake and body weight changes, but these need to be of sufficient duration. Carrying out a study with sufficient study duration, incorporating the necessary measures to reduce confounding factors while maintaining participant compliance, however, constitutes a significantly greater logistical endeavour than the mostly prospective observational trials and cross-sectional studies that have been employed to date.  In 2014, the results of the GLYNDIET trial were published. This randomized controlled dietary intervention trial lasted six months and had aimed to systematically address these major limitations. The study was carried out by researchers at the Human Nutrition Unit, Faculty of Medicine and Health Sciences, Institute of Health Pere Virgili at the University of Rovira i Virgili, and the University Hospital Sant Joan, in Reus, Spain, in collaboration with the Instituto de Salud Carlos III, Madrid, Spain. Participants were assigned to one of three groups, all receiving an isocaloric energy-restricted diet with the same content of fiber, but distinguished by the diets either containing carbohydrates with a low-glycemic index (n=41 participants), carbohydrates with a high-glycemic index (n=41), or a reduced fat content (n=40), respectively (Table 1). The study participants were non-diabetic overweight-obese adults (body mass index (BMI) of 27 to 35 kg/m2) between 30 and 60 years old. The applied energy restriction was 500 kcal of total dietary energy intake for all participants, with the total daily energy intake adjusted for body weight according to four groups of body weight ranges. The groups were balanced for gender (~24% women) and age (mean age of ~45 yrs). No differences in mean values of baseline clinical characteristics were apparent between groups, including average fasting-plasma glucose levels of ~100 mg/dl, indicating that participants were normoglycemic. There was a small difference in energy from protein intake (18.8%, 18.4% and 17.0%, respectively). Adherence to the diets was executed by registered dietitians providing specific recommendations, in particular on the type of carbohydrates and cooking methods used. Biweekly menus and recipes were provided to promote adherence. Energy and nutrient intake was calculated using a food composition table for Spain. Several participants did not complete the study, with 36, 37 and 31 persons in the above-mentioned study groups finishing the study, respectively.
In 2014, the results of the GLYNDIET trial were published. This randomized controlled dietary intervention trial lasted six months and had aimed to systematically address these major limitations. The study was carried out by researchers at the Human Nutrition Unit, Faculty of Medicine and Health Sciences, Institute of Health Pere Virgili at the University of Rovira i Virgili, and the University Hospital Sant Joan, in Reus, Spain, in collaboration with the Instituto de Salud Carlos III, Madrid, Spain. Participants were assigned to one of three groups, all receiving an isocaloric energy-restricted diet with the same content of fiber, but distinguished by the diets either containing carbohydrates with a low-glycemic index (n=41 participants), carbohydrates with a high-glycemic index (n=41), or a reduced fat content (n=40), respectively (Table 1). The study participants were non-diabetic overweight-obese adults (body mass index (BMI) of 27 to 35 kg/m2) between 30 and 60 years old. The applied energy restriction was 500 kcal of total dietary energy intake for all participants, with the total daily energy intake adjusted for body weight according to four groups of body weight ranges. The groups were balanced for gender (~24% women) and age (mean age of ~45 yrs). No differences in mean values of baseline clinical characteristics were apparent between groups, including average fasting-plasma glucose levels of ~100 mg/dl, indicating that participants were normoglycemic. There was a small difference in energy from protein intake (18.8%, 18.4% and 17.0%, respectively). Adherence to the diets was executed by registered dietitians providing specific recommendations, in particular on the type of carbohydrates and cooking methods used. Biweekly menus and recipes were provided to promote adherence. Energy and nutrient intake was calculated using a food composition table for Spain. Several participants did not complete the study, with 36, 37 and 31 persons in the above-mentioned study groups finishing the study, respectively. 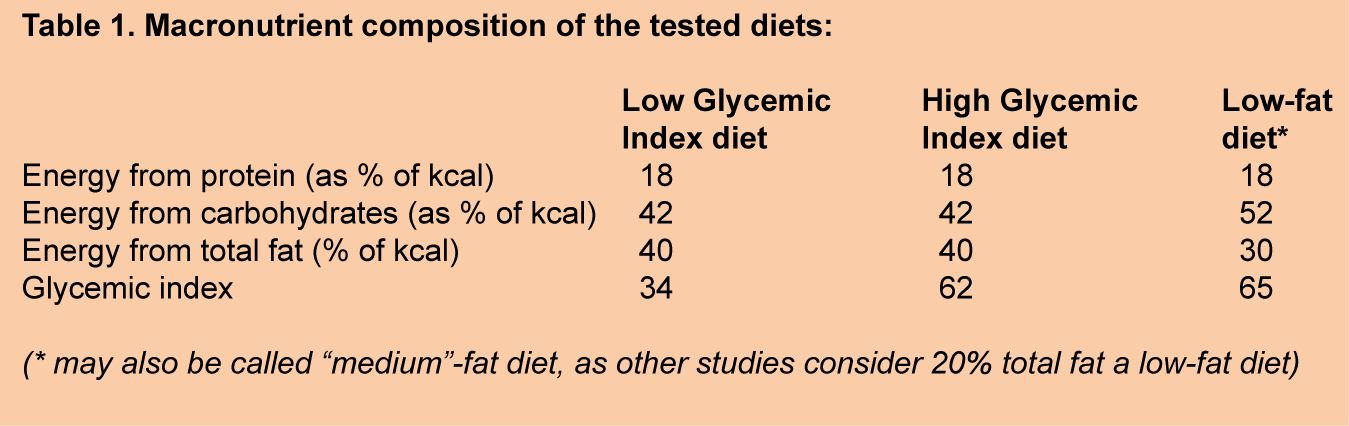 The results of the GLYNDIET study showed that the decrease in BMI in participants following the low-glycemic index diet was greater than with the low-fat diet (2.5 vs 1.5 kg/m2 decrease in BMI, respectively). The decrease in BMI in people receiving a high-glycemic index diet was borderline significant (P=0.061) compared to the low-fat diet, and comparable to that achieved with the low-glycemic index diet. Decreases in insulin resistance and improvements in beta-cell function were significantly greater in the low-glycemic index diet than the other two diets. Participants on the low-glycemic index diet also reported feeling less hungry when challenged to a breakfast test compared to those on the high-glycemic index diet. With the three diets achieving weight loss, and the low-glycemic index diet having a more favorable effect on glucose regulation, one aspect that had not been addressed was if the distinct differences in macronutrients would affect other metabolic aspects of weight loss. While the effect of dietary fat quality on fatty acid composition of tissue membranes has been studied extensively, the effect of foods with different carbohydrate qualities is virtually undocumented.
The results of the GLYNDIET study showed that the decrease in BMI in participants following the low-glycemic index diet was greater than with the low-fat diet (2.5 vs 1.5 kg/m2 decrease in BMI, respectively). The decrease in BMI in people receiving a high-glycemic index diet was borderline significant (P=0.061) compared to the low-fat diet, and comparable to that achieved with the low-glycemic index diet. Decreases in insulin resistance and improvements in beta-cell function were significantly greater in the low-glycemic index diet than the other two diets. Participants on the low-glycemic index diet also reported feeling less hungry when challenged to a breakfast test compared to those on the high-glycemic index diet. With the three diets achieving weight loss, and the low-glycemic index diet having a more favorable effect on glucose regulation, one aspect that had not been addressed was if the distinct differences in macronutrients would affect other metabolic aspects of weight loss. While the effect of dietary fat quality on fatty acid composition of tissue membranes has been studied extensively, the effect of foods with different carbohydrate qualities is virtually undocumented.  In the present study, Giardina and colleagues report on the effects of the three energy-restricted diets with different macronutrient compositions studied in the GLYNDIET trial on fatty acid composition. The analysis was carried out by the original GLYNDIET researchers together with colleagues from the Lipid Clinic at the Hospital Clínic, Institut d’Investigacions Biomèdiques August Pi I Sunyer, in Barcelona. Blood samples had been taken after an overnight fast at the start of the study and at the end of the 6-month intervention, and washed red blood cells (RBCs) prepared from EDTA plasma had been collected. Fatty acid profiles were determined by gas chromatography-flame ionization detection of fatty acid-methyl esters, following trans-methylation of membranes prepared from lysed RBCs. No information was provided on how long after completion of the GLYNDIET trial the RBC fatty acid profiles were determined, or on how samples were stored. Results on fatty acid levels in RBCs were available from 30, 31 and 26 participants of the high-glycemic index, low-glycemic index, and low-fat diet groups, respectively. These study participants showed similar characteristics to those of the overall study cohort. In the participants for which fatty acid results were available, the low-glycemic and high-glycemic index diets promoted greater weight loss than the low-fat diet (7.4, 7.2 and 4.4 kg, respectively). Participants in the low-glycemic index group displayed a significantly higher dietary intake of polyunsaturated fatty acids (~30% increase) over the study period. The assessment of the dietary intakes of individual PUFA species revealed no significant changes in intakes of eicosapentaenoic acid (EPA), docosahexaenoic acid (DHA), alpha-linolenic acid (ALA), total omega-3 PUFA or total omega-6 PUFA between groups. Higher baseline intake of EPA and DHA in the low-fat diet group suggested that groups were not completely matched at baseline, or that the precise dietary intake of individual fatty acids was challenging to estimate from food records. Fiber intake was significantly decreased in the low-glycemic index group.
In the present study, Giardina and colleagues report on the effects of the three energy-restricted diets with different macronutrient compositions studied in the GLYNDIET trial on fatty acid composition. The analysis was carried out by the original GLYNDIET researchers together with colleagues from the Lipid Clinic at the Hospital Clínic, Institut d’Investigacions Biomèdiques August Pi I Sunyer, in Barcelona. Blood samples had been taken after an overnight fast at the start of the study and at the end of the 6-month intervention, and washed red blood cells (RBCs) prepared from EDTA plasma had been collected. Fatty acid profiles were determined by gas chromatography-flame ionization detection of fatty acid-methyl esters, following trans-methylation of membranes prepared from lysed RBCs. No information was provided on how long after completion of the GLYNDIET trial the RBC fatty acid profiles were determined, or on how samples were stored. Results on fatty acid levels in RBCs were available from 30, 31 and 26 participants of the high-glycemic index, low-glycemic index, and low-fat diet groups, respectively. These study participants showed similar characteristics to those of the overall study cohort. In the participants for which fatty acid results were available, the low-glycemic and high-glycemic index diets promoted greater weight loss than the low-fat diet (7.4, 7.2 and 4.4 kg, respectively). Participants in the low-glycemic index group displayed a significantly higher dietary intake of polyunsaturated fatty acids (~30% increase) over the study period. The assessment of the dietary intakes of individual PUFA species revealed no significant changes in intakes of eicosapentaenoic acid (EPA), docosahexaenoic acid (DHA), alpha-linolenic acid (ALA), total omega-3 PUFA or total omega-6 PUFA between groups. Higher baseline intake of EPA and DHA in the low-fat diet group suggested that groups were not completely matched at baseline, or that the precise dietary intake of individual fatty acids was challenging to estimate from food records. Fiber intake was significantly decreased in the low-glycemic index group.  Objective values of LCPUFA resulting from longer-term dietary intake can be obtained from measuring their levels in RBC membranes. After six months, significant decreases in EPA, DHA and total omega-3 LCPUFA level in RBC membranes (20%, 8.2% and 8.0%, respectively), as well as in the omega-3 index (0.5% point decrease from a baseline omega-3 index of 6.9%), were observed in the participants of the low-fat diet group. These changes were not observed in the high- and low-glycemic index groups, except a 13% decrease in EPA level in the high-glycemic index group. In the low-fat diet group, the levels of the omega-6 LCPUFA adrenic acid (C22:4 n-6) and docosapentaenoic acid (C22:5 n-6) increased over the study period (6.7 % and 20%, respectively), a change not seen in in the other study groups. Reductions in ALA levels were observed in all groups, although the baseline values were low. The present study is among the first to indicate that energy restriction on a low-glycemic index diet does not affect LCPUFA status (at the achieved level of weight loss in the study). Although weight-loss can be achieved employing diverse diets with different macronutrient composition, a reduction in total dietary fat content had a significantly more marked effect on endogenous LCPUFA status than modifying the carbohydrate quality of the diet. The results of this study may contribute to a optimization of the weight-loss approach which is to be used for an overweight person.
Objective values of LCPUFA resulting from longer-term dietary intake can be obtained from measuring their levels in RBC membranes. After six months, significant decreases in EPA, DHA and total omega-3 LCPUFA level in RBC membranes (20%, 8.2% and 8.0%, respectively), as well as in the omega-3 index (0.5% point decrease from a baseline omega-3 index of 6.9%), were observed in the participants of the low-fat diet group. These changes were not observed in the high- and low-glycemic index groups, except a 13% decrease in EPA level in the high-glycemic index group. In the low-fat diet group, the levels of the omega-6 LCPUFA adrenic acid (C22:4 n-6) and docosapentaenoic acid (C22:5 n-6) increased over the study period (6.7 % and 20%, respectively), a change not seen in in the other study groups. Reductions in ALA levels were observed in all groups, although the baseline values were low. The present study is among the first to indicate that energy restriction on a low-glycemic index diet does not affect LCPUFA status (at the achieved level of weight loss in the study). Although weight-loss can be achieved employing diverse diets with different macronutrient composition, a reduction in total dietary fat content had a significantly more marked effect on endogenous LCPUFA status than modifying the carbohydrate quality of the diet. The results of this study may contribute to a optimization of the weight-loss approach which is to be used for an overweight person.  Recent studies have provided marked new insight that no single weight loss approach fits every individual, with fasting glycaemia and insulin sensitivity predicting an individual’s potential for weight loss or gain for different types of diets. Normoglycemic individuals appear to benefit more from an energy-restricted low-fat high-carbohydrate diet for losing weight, whereas pre-diabetic and diabetic individuals will lose weight on diets characterized by a high-fat low-carbohydrate diet or focusing on carbohydrate intakes with a low glycemic index, high fiber content and whole grain consumption. Maintaining a reduced bodyweight, once achieved, also depends on glycemic load, with even further weight loss possible in prediabetic people with a low glycemic load maintenance diet. Maintenance of the achieved lower body weight is sustainable in normoglycemic persons using this approach. The results of the present study also suggest that overweight-obese individuals who do not yet show signs of diabetes and who aim to lose weight through a reduced fat intake, should consider additional intake of omega-3 LCPUFA in order to maintain sufficient dietary levels of these fatty acids. On the other hand, the study indicates that fiber intake may also need to be corrected in individuals losing weight on a low-glycemic index diet. A limitation of the current study is that the results are restricted to the population in which these were obtained, namely in Spain. The average omega-3 index is high, with one study reporting a value of 9.1% in the Spanish population. EPA+DHA intake clearly contributes to high RBC levels of EPA and DHA in this population, but only partly explains why the levels are relatively high, and other (unknown) environmental and dietary factors are likely contributing as well. The omega-3 index of the GLYNDIET participants was lower (~6.8%) and did not change during the 6-month weight loss intervention, except on the background of a low-fat diet, when the omega-3 LCPUFA status decreased. Further attention needs to be paid to the observation that ALA levels decreased in all tested weight loss protocols. The latter may relate to the fact that the Spanish population has a relatively high vegetable intake, and a lower energy intake overall may lead to reduced ALA-intake. A further limitation of this study relates to the estimation of fatty acid intake based on 3-day food records, which may significantly underestimate or skew the estimated intake of specific fatty acids that are infrequently consumed. Future studies that employ longer food records may possibly provide more reliable dietary intake estimates for specific fatty acids. It needs to be acknowledged that it is essentially impossible to alter any aspect of the diet without affecting something else, including the composition of fatty acids in the diet. Whereas olive oil was the main reported source of dietary fat in the low- and high-glycemic index diets, dairy was a main source of fat in the low fat diet group. Therefore, it remains a possibility that the changes in endogenous FA profile associated with the low fat diet could have been due to changes in the composition of ingested fatty acids, and which were not detected by diet records due to their lack of sensitivity.
Recent studies have provided marked new insight that no single weight loss approach fits every individual, with fasting glycaemia and insulin sensitivity predicting an individual’s potential for weight loss or gain for different types of diets. Normoglycemic individuals appear to benefit more from an energy-restricted low-fat high-carbohydrate diet for losing weight, whereas pre-diabetic and diabetic individuals will lose weight on diets characterized by a high-fat low-carbohydrate diet or focusing on carbohydrate intakes with a low glycemic index, high fiber content and whole grain consumption. Maintaining a reduced bodyweight, once achieved, also depends on glycemic load, with even further weight loss possible in prediabetic people with a low glycemic load maintenance diet. Maintenance of the achieved lower body weight is sustainable in normoglycemic persons using this approach. The results of the present study also suggest that overweight-obese individuals who do not yet show signs of diabetes and who aim to lose weight through a reduced fat intake, should consider additional intake of omega-3 LCPUFA in order to maintain sufficient dietary levels of these fatty acids. On the other hand, the study indicates that fiber intake may also need to be corrected in individuals losing weight on a low-glycemic index diet. A limitation of the current study is that the results are restricted to the population in which these were obtained, namely in Spain. The average omega-3 index is high, with one study reporting a value of 9.1% in the Spanish population. EPA+DHA intake clearly contributes to high RBC levels of EPA and DHA in this population, but only partly explains why the levels are relatively high, and other (unknown) environmental and dietary factors are likely contributing as well. The omega-3 index of the GLYNDIET participants was lower (~6.8%) and did not change during the 6-month weight loss intervention, except on the background of a low-fat diet, when the omega-3 LCPUFA status decreased. Further attention needs to be paid to the observation that ALA levels decreased in all tested weight loss protocols. The latter may relate to the fact that the Spanish population has a relatively high vegetable intake, and a lower energy intake overall may lead to reduced ALA-intake. A further limitation of this study relates to the estimation of fatty acid intake based on 3-day food records, which may significantly underestimate or skew the estimated intake of specific fatty acids that are infrequently consumed. Future studies that employ longer food records may possibly provide more reliable dietary intake estimates for specific fatty acids. It needs to be acknowledged that it is essentially impossible to alter any aspect of the diet without affecting something else, including the composition of fatty acids in the diet. Whereas olive oil was the main reported source of dietary fat in the low- and high-glycemic index diets, dairy was a main source of fat in the low fat diet group. Therefore, it remains a possibility that the changes in endogenous FA profile associated with the low fat diet could have been due to changes in the composition of ingested fatty acids, and which were not detected by diet records due to their lack of sensitivity.  Weight loss itself has been reported to be associated with changes in RBC fatty acid composition. In a small study, weight loss following subtotal gastrectomy markedly improved DHA tissue levels and omega-3 LCPUFA biosynthesis capacity of obese patients. A strong influence of body weight on omega-3 LCPUFA status following supplementation has also been recently uncovered in young people. The current results indicate that body weight reduction by itself was, however, not the main driver of changes in fatty acid composition, but rather that weight loss achieved by a low-fat diet may be associated with reductions in omega-3 LCPUFA, whereas a change in the type of carbohydrates during weight loss did not have such marked effects on LCPUFA status in non-diabetic overweight-obese adults. This study has provided a useful insight into the effect of different macronutrient compositions that can be provided during a weight loss schedule. Further studies that reduce confounding factors as much as possible may improve the already available approaches for achieving weight loss in individuals and different medical conditions, by minimizing the potential adverse effects on essential nutrient intake and their endogenous levels. Giardina S, Sala-Vila A, Hernández-Alonso P, Calvo C, Salas-Salvadó J, Bulló M. Carbohydrate quality and quantity affects the composition of the red blood cell fatty acid membrane in overweight and obese individuals. Clin. Nutr. 2017;Feb 11. [PubMed] Worth Noting Astrup A, Hjorth MF. Low-fat or low carb for weight loss? It depends on your glucose metabolism. EBioMedicine 2017. [PubMed] Christian LM, Young AS, Mitchell AM, Belury MA, Gracious BL, Arnold LE, Fristad MA. Body weight affects omega-3 polyunsaturated fatty acid (PUFA) accumulation in youth following supplementation in post-hoc analyses of a randomized controlled trial. PLoS One 2017;12(4):e0173087. [PubMed] Elizondo A, Araya J, Rodrigo R, Signorini C, Sgherri C, Comporti M, Poniachik J, Videla LA. Effects of weight loss on liver and erythrocyte polyunsaturated fatty acid pattern and oxidative stress status in obese patients with non-alcoholic fatty liver disease. Biol. Res. 2008;41(1):59-68. [PubMed] Fasting plasma glucose values: American Diabetes Association, Clinical Diabetes, 18, 2, 2000. [Link] Global Burden of Disease 2015 Obesity Collaborators, Health Effects of Overweight and Obesity in 195 Countries over 25 Years. N Engl J Med 2017;377(1):13-27. [PubMed] Gregg EW, Shaw JE. Global health effects of overweight and obesity. N. Engl. J. Med. 2017;377(1):80-81. [PubMed] Hjorth MF, Ritz C, Blaak EE, Saris WH, Langin D, Poulsen SK, Larsen TM, Sørensen TI, Zohar Y, Astrup A. Pretreatment fasting plasma glucose and insulin modify dietary weight loss success: results from 3 randomized clinical trials. Am. J. Clin. Nutr. 2017;July 5. [PubMed] Juanola-Falgarona M, Ibarrola-Jurado N, Salas-Salvadó J, Rabassa-Soler A, Bulló M. Design and methods of the GLYNDIET study; assessing the role of glycemic index on weight loss and metabolic risk markers. Nutr. Hosp. 2013;28(2):382-390. [PubMed] Juanola-Falgarona M, Salas-Salvado J, Ibarrola-Jurado N, Rabassa-Soler A, Diaz-Lopez A, Guasch-Ferre M, Hernandez-Alonso P, Balanza R, Bullo M. Effect of the glycemic index of the diet on weight loss, modulation of satiety, inflammation, and other metabolic risk factors: a randomized controlled trial. Am. J. Clin. Nutr. 2014;100(1):27-35. [PubMed] Mataix-Verdú J, Mañas-Almendros M, J. L-G, Martínez de Victoria-Muñoz E, Sänchez JJ, Borregón-Martínez A. Tabla de composición de alimentos españoles. Editorial Universidad de Granada. 2013; Campus Universitario de Cartuja.(5th ed.). [Link] Meinhold CL. Low-glycemic load diets: How does the evidence for prevention of disease measure up? J. Am. Diet Assoc. 2010;110(12):1818-1819. [PubMed] Meinhold CL, Dodd KW, Jiao L, Flood A, Shikany JM, Genkinger JM, Hayes RB, Stolzenberg-Solomon RZ. Available carbohydrates, glycemic load, and pancreatic cancer: is there a link? Am. J. Epidemiol. 2010;171(11):1174-1182. [PubMed] Prospective Studies Collaboration, Whitlock G, Lewington S, Sherliker P, Clarke R, Emberson J, Halsey J, Qizilbash N, Collins R, Peto R. Body-mass index and cause-specific mortality in 900 000 adults: collaborative analyses of 57 prospective studies. Lancet 2009;373(9669):1083-1096. [PubMed] Sala-Vila A, Harris WS, Cofán M, Pérez-Heras AM, Pintó X, Lamuela-Raventós RM, Covas MI, Estruch R, Ros E. Determinants of the omega-3 index in a Mediterranean population at increased risk for CHD. Br. J. Nutr. 2011;106(3):425-431. [PubMed]
Weight loss itself has been reported to be associated with changes in RBC fatty acid composition. In a small study, weight loss following subtotal gastrectomy markedly improved DHA tissue levels and omega-3 LCPUFA biosynthesis capacity of obese patients. A strong influence of body weight on omega-3 LCPUFA status following supplementation has also been recently uncovered in young people. The current results indicate that body weight reduction by itself was, however, not the main driver of changes in fatty acid composition, but rather that weight loss achieved by a low-fat diet may be associated with reductions in omega-3 LCPUFA, whereas a change in the type of carbohydrates during weight loss did not have such marked effects on LCPUFA status in non-diabetic overweight-obese adults. This study has provided a useful insight into the effect of different macronutrient compositions that can be provided during a weight loss schedule. Further studies that reduce confounding factors as much as possible may improve the already available approaches for achieving weight loss in individuals and different medical conditions, by minimizing the potential adverse effects on essential nutrient intake and their endogenous levels. Giardina S, Sala-Vila A, Hernández-Alonso P, Calvo C, Salas-Salvadó J, Bulló M. Carbohydrate quality and quantity affects the composition of the red blood cell fatty acid membrane in overweight and obese individuals. Clin. Nutr. 2017;Feb 11. [PubMed] Worth Noting Astrup A, Hjorth MF. Low-fat or low carb for weight loss? It depends on your glucose metabolism. EBioMedicine 2017. [PubMed] Christian LM, Young AS, Mitchell AM, Belury MA, Gracious BL, Arnold LE, Fristad MA. Body weight affects omega-3 polyunsaturated fatty acid (PUFA) accumulation in youth following supplementation in post-hoc analyses of a randomized controlled trial. PLoS One 2017;12(4):e0173087. [PubMed] Elizondo A, Araya J, Rodrigo R, Signorini C, Sgherri C, Comporti M, Poniachik J, Videla LA. Effects of weight loss on liver and erythrocyte polyunsaturated fatty acid pattern and oxidative stress status in obese patients with non-alcoholic fatty liver disease. Biol. Res. 2008;41(1):59-68. [PubMed] Fasting plasma glucose values: American Diabetes Association, Clinical Diabetes, 18, 2, 2000. [Link] Global Burden of Disease 2015 Obesity Collaborators, Health Effects of Overweight and Obesity in 195 Countries over 25 Years. N Engl J Med 2017;377(1):13-27. [PubMed] Gregg EW, Shaw JE. Global health effects of overweight and obesity. N. Engl. J. Med. 2017;377(1):80-81. [PubMed] Hjorth MF, Ritz C, Blaak EE, Saris WH, Langin D, Poulsen SK, Larsen TM, Sørensen TI, Zohar Y, Astrup A. Pretreatment fasting plasma glucose and insulin modify dietary weight loss success: results from 3 randomized clinical trials. Am. J. Clin. Nutr. 2017;July 5. [PubMed] Juanola-Falgarona M, Ibarrola-Jurado N, Salas-Salvadó J, Rabassa-Soler A, Bulló M. Design and methods of the GLYNDIET study; assessing the role of glycemic index on weight loss and metabolic risk markers. Nutr. Hosp. 2013;28(2):382-390. [PubMed] Juanola-Falgarona M, Salas-Salvado J, Ibarrola-Jurado N, Rabassa-Soler A, Diaz-Lopez A, Guasch-Ferre M, Hernandez-Alonso P, Balanza R, Bullo M. Effect of the glycemic index of the diet on weight loss, modulation of satiety, inflammation, and other metabolic risk factors: a randomized controlled trial. Am. J. Clin. Nutr. 2014;100(1):27-35. [PubMed] Mataix-Verdú J, Mañas-Almendros M, J. L-G, Martínez de Victoria-Muñoz E, Sänchez JJ, Borregón-Martínez A. Tabla de composición de alimentos españoles. Editorial Universidad de Granada. 2013; Campus Universitario de Cartuja.(5th ed.). [Link] Meinhold CL. Low-glycemic load diets: How does the evidence for prevention of disease measure up? J. Am. Diet Assoc. 2010;110(12):1818-1819. [PubMed] Meinhold CL, Dodd KW, Jiao L, Flood A, Shikany JM, Genkinger JM, Hayes RB, Stolzenberg-Solomon RZ. Available carbohydrates, glycemic load, and pancreatic cancer: is there a link? Am. J. Epidemiol. 2010;171(11):1174-1182. [PubMed] Prospective Studies Collaboration, Whitlock G, Lewington S, Sherliker P, Clarke R, Emberson J, Halsey J, Qizilbash N, Collins R, Peto R. Body-mass index and cause-specific mortality in 900 000 adults: collaborative analyses of 57 prospective studies. Lancet 2009;373(9669):1083-1096. [PubMed] Sala-Vila A, Harris WS, Cofán M, Pérez-Heras AM, Pintó X, Lamuela-Raventós RM, Covas MI, Estruch R, Ros E. Determinants of the omega-3 index in a Mediterranean population at increased risk for CHD. Br. J. Nutr. 2011;106(3):425-431. [PubMed]
- The influence on tissue fatty acid composition of three different isocaloric diets containing carbohydrates with a low glycemic index, carbohydrates with a high glycemic index, or a reduced fat content, respectively, was assessed in overweight-obese adults on a 500 kcal/d-energy restriction in a randomized controlled intervention trial.
- The study had been designed to avoid previous limitations in addressing the effect of macronutrients on weight loss, and revealed improvements in insulin control in participants on the low-glycemic index diet. However, potential adverse effects on polyunsaturated fatty acid (PUFA) status had not been previously investigated.
- After six months, no changes in red blood cell membrane fatty acid composition were observed in participants on the low-glycemic index diet, eicosapentaenoic acid (EPA) level decreased in the high-glycemic index group, and decreases in several omega-3 LCPUFAs and increases in omega-6 LCPUFAs were observed in the low-fat diet group.
- This study suggests that during energy-restricted weight loss PUFA levels are sensitive to changes in total dietary fat content, but are not perturbed, or much less affected, by diets with varying carbohydrate profile.
 The global obesity epidemic is still getting worse in most parts of the world, as highlighted in recent reports. Addressing the obesity problem is important because being overweight is associated with increased mortality from a range of metabolic disorders, but on the other hand it potentially can be addressed. Obesity-associated disease is not uniform across populations, stimulating interest in understanding how weight gain prevention and promotion of weight loss can be best addressed. An unresolved issue is whether chronic excessive intake of easily available carbohydrates promotes obesity prevalence. Similarly, it is important to know if lowering the intake of carbohydrates in overweight individuals can contribute to weight loss. Further, with carbohydrates being important macronutrients, would lowering their intake in overweight people have any unintended effects? Hyperglycemia and hyperinsulinemia following chronically-elevated dietary intake of carbohydrates are responsible for increased fat storage and insulin resistance due to accelerated oxidation of circulating glucose to reactive advanced glycation end-products in vascular tissues. The inflammation of metabolically-active tissues has also been implicated in dysregulated metabolic responses to elevated carbohydrate intake. In contrast with these hypotheses, however, are views that indicate that these mechanisms are not of sufficient magnitude or duration to modify the processes the body uses to handle excess energy intake or sugars specifically. Furthermore, much of the evidence linking carbohydrate intake and weight gain is based on observational and cross-sectional studies, whereas intervention studies that could provide a more definite answer have been small in sample size, of too short duration, or did not control for potential confounders. Although intuitively a simple relationship, at present it is still a leap to conclude that a diet with a low glycemic load can prevent weight gain or significantly contribute to weight loss. Several methodological limitations need to be overcome to allow drawing solid conclusions on the relationship between carbohydrate intake, glycemic index and load, and weight gain or loss. For a start, prospective studies need to adequately monitor food intake. Food records are preferred over the use of food-frequency questionnaires that rely on people’s (generally poor) memories of past dietary habits, and cognitive ability. Food records also allow a better recording of the specific details on how food was prepared, which affects the glycemic index and load of food items rich in carbohydrates. However, maintaining a food record by study participants carries a risk that dietary habits may change, generally towards currently fashionable dietary recommendations. Furthermore, measurement error should be eliminated through professional supervision guiding participants on how to fill out their records. Ideally, multiple dietary records should be taken that span a sufficiently long period and cover the different seasons in studies lasting more than a year.
The global obesity epidemic is still getting worse in most parts of the world, as highlighted in recent reports. Addressing the obesity problem is important because being overweight is associated with increased mortality from a range of metabolic disorders, but on the other hand it potentially can be addressed. Obesity-associated disease is not uniform across populations, stimulating interest in understanding how weight gain prevention and promotion of weight loss can be best addressed. An unresolved issue is whether chronic excessive intake of easily available carbohydrates promotes obesity prevalence. Similarly, it is important to know if lowering the intake of carbohydrates in overweight individuals can contribute to weight loss. Further, with carbohydrates being important macronutrients, would lowering their intake in overweight people have any unintended effects? Hyperglycemia and hyperinsulinemia following chronically-elevated dietary intake of carbohydrates are responsible for increased fat storage and insulin resistance due to accelerated oxidation of circulating glucose to reactive advanced glycation end-products in vascular tissues. The inflammation of metabolically-active tissues has also been implicated in dysregulated metabolic responses to elevated carbohydrate intake. In contrast with these hypotheses, however, are views that indicate that these mechanisms are not of sufficient magnitude or duration to modify the processes the body uses to handle excess energy intake or sugars specifically. Furthermore, much of the evidence linking carbohydrate intake and weight gain is based on observational and cross-sectional studies, whereas intervention studies that could provide a more definite answer have been small in sample size, of too short duration, or did not control for potential confounders. Although intuitively a simple relationship, at present it is still a leap to conclude that a diet with a low glycemic load can prevent weight gain or significantly contribute to weight loss. Several methodological limitations need to be overcome to allow drawing solid conclusions on the relationship between carbohydrate intake, glycemic index and load, and weight gain or loss. For a start, prospective studies need to adequately monitor food intake. Food records are preferred over the use of food-frequency questionnaires that rely on people’s (generally poor) memories of past dietary habits, and cognitive ability. Food records also allow a better recording of the specific details on how food was prepared, which affects the glycemic index and load of food items rich in carbohydrates. However, maintaining a food record by study participants carries a risk that dietary habits may change, generally towards currently fashionable dietary recommendations. Furthermore, measurement error should be eliminated through professional supervision guiding participants on how to fill out their records. Ideally, multiple dietary records should be taken that span a sufficiently long period and cover the different seasons in studies lasting more than a year.  A well-recognized limitation of observational studies in evaluating the association of individual food nutrients with outcomes is the need to control for many other variables that co-vary with the nutrient of interest. However, randomized controlled studies may also suffer from this limitation if the assigned diets vary in dietary components that are not being controlled. For example, a diet aiming to evaluate low-glycemic index carbohydrates may have exchanged one food component with another containing less fiber or with a different amount of protein. All macronutrient classes, as well as micronutrients, should ideally be controlled in order to draw meaningful conclusions. Observational studies are generally limited to establishing associations. These are not causal to the endpoint, and sometimes reversed causation is at play where the outcome affects the intake of the nutrient being investigated. For example, in an observational trial that lasted 6.5 years, an increase in pancreatic cancer risk associated with high available carbohydrate and low fat intake was thought to reflect the dietary changes that are associated with subclinical disease. Controlled intervention studies are preferred when intending to dissect the relationship between carbohydrate intake and body weight changes, but these need to be of sufficient duration. Carrying out a study with sufficient study duration, incorporating the necessary measures to reduce confounding factors while maintaining participant compliance, however, constitutes a significantly greater logistical endeavour than the mostly prospective observational trials and cross-sectional studies that have been employed to date.
A well-recognized limitation of observational studies in evaluating the association of individual food nutrients with outcomes is the need to control for many other variables that co-vary with the nutrient of interest. However, randomized controlled studies may also suffer from this limitation if the assigned diets vary in dietary components that are not being controlled. For example, a diet aiming to evaluate low-glycemic index carbohydrates may have exchanged one food component with another containing less fiber or with a different amount of protein. All macronutrient classes, as well as micronutrients, should ideally be controlled in order to draw meaningful conclusions. Observational studies are generally limited to establishing associations. These are not causal to the endpoint, and sometimes reversed causation is at play where the outcome affects the intake of the nutrient being investigated. For example, in an observational trial that lasted 6.5 years, an increase in pancreatic cancer risk associated with high available carbohydrate and low fat intake was thought to reflect the dietary changes that are associated with subclinical disease. Controlled intervention studies are preferred when intending to dissect the relationship between carbohydrate intake and body weight changes, but these need to be of sufficient duration. Carrying out a study with sufficient study duration, incorporating the necessary measures to reduce confounding factors while maintaining participant compliance, however, constitutes a significantly greater logistical endeavour than the mostly prospective observational trials and cross-sectional studies that have been employed to date.  In 2014, the results of the GLYNDIET trial were published. This randomized controlled dietary intervention trial lasted six months and had aimed to systematically address these major limitations. The study was carried out by researchers at the Human Nutrition Unit, Faculty of Medicine and Health Sciences, Institute of Health Pere Virgili at the University of Rovira i Virgili, and the University Hospital Sant Joan, in Reus, Spain, in collaboration with the Instituto de Salud Carlos III, Madrid, Spain. Participants were assigned to one of three groups, all receiving an isocaloric energy-restricted diet with the same content of fiber, but distinguished by the diets either containing carbohydrates with a low-glycemic index (n=41 participants), carbohydrates with a high-glycemic index (n=41), or a reduced fat content (n=40), respectively (Table 1). The study participants were non-diabetic overweight-obese adults (body mass index (BMI) of 27 to 35 kg/m2) between 30 and 60 years old. The applied energy restriction was 500 kcal of total dietary energy intake for all participants, with the total daily energy intake adjusted for body weight according to four groups of body weight ranges. The groups were balanced for gender (~24% women) and age (mean age of ~45 yrs). No differences in mean values of baseline clinical characteristics were apparent between groups, including average fasting-plasma glucose levels of ~100 mg/dl, indicating that participants were normoglycemic. There was a small difference in energy from protein intake (18.8%, 18.4% and 17.0%, respectively). Adherence to the diets was executed by registered dietitians providing specific recommendations, in particular on the type of carbohydrates and cooking methods used. Biweekly menus and recipes were provided to promote adherence. Energy and nutrient intake was calculated using a food composition table for Spain. Several participants did not complete the study, with 36, 37 and 31 persons in the above-mentioned study groups finishing the study, respectively.
In 2014, the results of the GLYNDIET trial were published. This randomized controlled dietary intervention trial lasted six months and had aimed to systematically address these major limitations. The study was carried out by researchers at the Human Nutrition Unit, Faculty of Medicine and Health Sciences, Institute of Health Pere Virgili at the University of Rovira i Virgili, and the University Hospital Sant Joan, in Reus, Spain, in collaboration with the Instituto de Salud Carlos III, Madrid, Spain. Participants were assigned to one of three groups, all receiving an isocaloric energy-restricted diet with the same content of fiber, but distinguished by the diets either containing carbohydrates with a low-glycemic index (n=41 participants), carbohydrates with a high-glycemic index (n=41), or a reduced fat content (n=40), respectively (Table 1). The study participants were non-diabetic overweight-obese adults (body mass index (BMI) of 27 to 35 kg/m2) between 30 and 60 years old. The applied energy restriction was 500 kcal of total dietary energy intake for all participants, with the total daily energy intake adjusted for body weight according to four groups of body weight ranges. The groups were balanced for gender (~24% women) and age (mean age of ~45 yrs). No differences in mean values of baseline clinical characteristics were apparent between groups, including average fasting-plasma glucose levels of ~100 mg/dl, indicating that participants were normoglycemic. There was a small difference in energy from protein intake (18.8%, 18.4% and 17.0%, respectively). Adherence to the diets was executed by registered dietitians providing specific recommendations, in particular on the type of carbohydrates and cooking methods used. Biweekly menus and recipes were provided to promote adherence. Energy and nutrient intake was calculated using a food composition table for Spain. Several participants did not complete the study, with 36, 37 and 31 persons in the above-mentioned study groups finishing the study, respectively.  The results of the GLYNDIET study showed that the decrease in BMI in participants following the low-glycemic index diet was greater than with the low-fat diet (2.5 vs 1.5 kg/m2 decrease in BMI, respectively). The decrease in BMI in people receiving a high-glycemic index diet was borderline significant (P=0.061) compared to the low-fat diet, and comparable to that achieved with the low-glycemic index diet. Decreases in insulin resistance and improvements in beta-cell function were significantly greater in the low-glycemic index diet than the other two diets. Participants on the low-glycemic index diet also reported feeling less hungry when challenged to a breakfast test compared to those on the high-glycemic index diet. With the three diets achieving weight loss, and the low-glycemic index diet having a more favorable effect on glucose regulation, one aspect that had not been addressed was if the distinct differences in macronutrients would affect other metabolic aspects of weight loss. While the effect of dietary fat quality on fatty acid composition of tissue membranes has been studied extensively, the effect of foods with different carbohydrate qualities is virtually undocumented.
The results of the GLYNDIET study showed that the decrease in BMI in participants following the low-glycemic index diet was greater than with the low-fat diet (2.5 vs 1.5 kg/m2 decrease in BMI, respectively). The decrease in BMI in people receiving a high-glycemic index diet was borderline significant (P=0.061) compared to the low-fat diet, and comparable to that achieved with the low-glycemic index diet. Decreases in insulin resistance and improvements in beta-cell function were significantly greater in the low-glycemic index diet than the other two diets. Participants on the low-glycemic index diet also reported feeling less hungry when challenged to a breakfast test compared to those on the high-glycemic index diet. With the three diets achieving weight loss, and the low-glycemic index diet having a more favorable effect on glucose regulation, one aspect that had not been addressed was if the distinct differences in macronutrients would affect other metabolic aspects of weight loss. While the effect of dietary fat quality on fatty acid composition of tissue membranes has been studied extensively, the effect of foods with different carbohydrate qualities is virtually undocumented.  In the present study, Giardina and colleagues report on the effects of the three energy-restricted diets with different macronutrient compositions studied in the GLYNDIET trial on fatty acid composition. The analysis was carried out by the original GLYNDIET researchers together with colleagues from the Lipid Clinic at the Hospital Clínic, Institut d’Investigacions Biomèdiques August Pi I Sunyer, in Barcelona. Blood samples had been taken after an overnight fast at the start of the study and at the end of the 6-month intervention, and washed red blood cells (RBCs) prepared from EDTA plasma had been collected. Fatty acid profiles were determined by gas chromatography-flame ionization detection of fatty acid-methyl esters, following trans-methylation of membranes prepared from lysed RBCs. No information was provided on how long after completion of the GLYNDIET trial the RBC fatty acid profiles were determined, or on how samples were stored. Results on fatty acid levels in RBCs were available from 30, 31 and 26 participants of the high-glycemic index, low-glycemic index, and low-fat diet groups, respectively. These study participants showed similar characteristics to those of the overall study cohort. In the participants for which fatty acid results were available, the low-glycemic and high-glycemic index diets promoted greater weight loss than the low-fat diet (7.4, 7.2 and 4.4 kg, respectively). Participants in the low-glycemic index group displayed a significantly higher dietary intake of polyunsaturated fatty acids (~30% increase) over the study period. The assessment of the dietary intakes of individual PUFA species revealed no significant changes in intakes of eicosapentaenoic acid (EPA), docosahexaenoic acid (DHA), alpha-linolenic acid (ALA), total omega-3 PUFA or total omega-6 PUFA between groups. Higher baseline intake of EPA and DHA in the low-fat diet group suggested that groups were not completely matched at baseline, or that the precise dietary intake of individual fatty acids was challenging to estimate from food records. Fiber intake was significantly decreased in the low-glycemic index group.
In the present study, Giardina and colleagues report on the effects of the three energy-restricted diets with different macronutrient compositions studied in the GLYNDIET trial on fatty acid composition. The analysis was carried out by the original GLYNDIET researchers together with colleagues from the Lipid Clinic at the Hospital Clínic, Institut d’Investigacions Biomèdiques August Pi I Sunyer, in Barcelona. Blood samples had been taken after an overnight fast at the start of the study and at the end of the 6-month intervention, and washed red blood cells (RBCs) prepared from EDTA plasma had been collected. Fatty acid profiles were determined by gas chromatography-flame ionization detection of fatty acid-methyl esters, following trans-methylation of membranes prepared from lysed RBCs. No information was provided on how long after completion of the GLYNDIET trial the RBC fatty acid profiles were determined, or on how samples were stored. Results on fatty acid levels in RBCs were available from 30, 31 and 26 participants of the high-glycemic index, low-glycemic index, and low-fat diet groups, respectively. These study participants showed similar characteristics to those of the overall study cohort. In the participants for which fatty acid results were available, the low-glycemic and high-glycemic index diets promoted greater weight loss than the low-fat diet (7.4, 7.2 and 4.4 kg, respectively). Participants in the low-glycemic index group displayed a significantly higher dietary intake of polyunsaturated fatty acids (~30% increase) over the study period. The assessment of the dietary intakes of individual PUFA species revealed no significant changes in intakes of eicosapentaenoic acid (EPA), docosahexaenoic acid (DHA), alpha-linolenic acid (ALA), total omega-3 PUFA or total omega-6 PUFA between groups. Higher baseline intake of EPA and DHA in the low-fat diet group suggested that groups were not completely matched at baseline, or that the precise dietary intake of individual fatty acids was challenging to estimate from food records. Fiber intake was significantly decreased in the low-glycemic index group.  Objective values of LCPUFA resulting from longer-term dietary intake can be obtained from measuring their levels in RBC membranes. After six months, significant decreases in EPA, DHA and total omega-3 LCPUFA level in RBC membranes (20%, 8.2% and 8.0%, respectively), as well as in the omega-3 index (0.5% point decrease from a baseline omega-3 index of 6.9%), were observed in the participants of the low-fat diet group. These changes were not observed in the high- and low-glycemic index groups, except a 13% decrease in EPA level in the high-glycemic index group. In the low-fat diet group, the levels of the omega-6 LCPUFA adrenic acid (C22:4 n-6) and docosapentaenoic acid (C22:5 n-6) increased over the study period (6.7 % and 20%, respectively), a change not seen in in the other study groups. Reductions in ALA levels were observed in all groups, although the baseline values were low. The present study is among the first to indicate that energy restriction on a low-glycemic index diet does not affect LCPUFA status (at the achieved level of weight loss in the study). Although weight-loss can be achieved employing diverse diets with different macronutrient composition, a reduction in total dietary fat content had a significantly more marked effect on endogenous LCPUFA status than modifying the carbohydrate quality of the diet. The results of this study may contribute to a optimization of the weight-loss approach which is to be used for an overweight person.
Objective values of LCPUFA resulting from longer-term dietary intake can be obtained from measuring their levels in RBC membranes. After six months, significant decreases in EPA, DHA and total omega-3 LCPUFA level in RBC membranes (20%, 8.2% and 8.0%, respectively), as well as in the omega-3 index (0.5% point decrease from a baseline omega-3 index of 6.9%), were observed in the participants of the low-fat diet group. These changes were not observed in the high- and low-glycemic index groups, except a 13% decrease in EPA level in the high-glycemic index group. In the low-fat diet group, the levels of the omega-6 LCPUFA adrenic acid (C22:4 n-6) and docosapentaenoic acid (C22:5 n-6) increased over the study period (6.7 % and 20%, respectively), a change not seen in in the other study groups. Reductions in ALA levels were observed in all groups, although the baseline values were low. The present study is among the first to indicate that energy restriction on a low-glycemic index diet does not affect LCPUFA status (at the achieved level of weight loss in the study). Although weight-loss can be achieved employing diverse diets with different macronutrient composition, a reduction in total dietary fat content had a significantly more marked effect on endogenous LCPUFA status than modifying the carbohydrate quality of the diet. The results of this study may contribute to a optimization of the weight-loss approach which is to be used for an overweight person.  Recent studies have provided marked new insight that no single weight loss approach fits every individual, with fasting glycaemia and insulin sensitivity predicting an individual’s potential for weight loss or gain for different types of diets. Normoglycemic individuals appear to benefit more from an energy-restricted low-fat high-carbohydrate diet for losing weight, whereas pre-diabetic and diabetic individuals will lose weight on diets characterized by a high-fat low-carbohydrate diet or focusing on carbohydrate intakes with a low glycemic index, high fiber content and whole grain consumption. Maintaining a reduced bodyweight, once achieved, also depends on glycemic load, with even further weight loss possible in prediabetic people with a low glycemic load maintenance diet. Maintenance of the achieved lower body weight is sustainable in normoglycemic persons using this approach. The results of the present study also suggest that overweight-obese individuals who do not yet show signs of diabetes and who aim to lose weight through a reduced fat intake, should consider additional intake of omega-3 LCPUFA in order to maintain sufficient dietary levels of these fatty acids. On the other hand, the study indicates that fiber intake may also need to be corrected in individuals losing weight on a low-glycemic index diet. A limitation of the current study is that the results are restricted to the population in which these were obtained, namely in Spain. The average omega-3 index is high, with one study reporting a value of 9.1% in the Spanish population. EPA+DHA intake clearly contributes to high RBC levels of EPA and DHA in this population, but only partly explains why the levels are relatively high, and other (unknown) environmental and dietary factors are likely contributing as well. The omega-3 index of the GLYNDIET participants was lower (~6.8%) and did not change during the 6-month weight loss intervention, except on the background of a low-fat diet, when the omega-3 LCPUFA status decreased. Further attention needs to be paid to the observation that ALA levels decreased in all tested weight loss protocols. The latter may relate to the fact that the Spanish population has a relatively high vegetable intake, and a lower energy intake overall may lead to reduced ALA-intake. A further limitation of this study relates to the estimation of fatty acid intake based on 3-day food records, which may significantly underestimate or skew the estimated intake of specific fatty acids that are infrequently consumed. Future studies that employ longer food records may possibly provide more reliable dietary intake estimates for specific fatty acids. It needs to be acknowledged that it is essentially impossible to alter any aspect of the diet without affecting something else, including the composition of fatty acids in the diet. Whereas olive oil was the main reported source of dietary fat in the low- and high-glycemic index diets, dairy was a main source of fat in the low fat diet group. Therefore, it remains a possibility that the changes in endogenous FA profile associated with the low fat diet could have been due to changes in the composition of ingested fatty acids, and which were not detected by diet records due to their lack of sensitivity.
Recent studies have provided marked new insight that no single weight loss approach fits every individual, with fasting glycaemia and insulin sensitivity predicting an individual’s potential for weight loss or gain for different types of diets. Normoglycemic individuals appear to benefit more from an energy-restricted low-fat high-carbohydrate diet for losing weight, whereas pre-diabetic and diabetic individuals will lose weight on diets characterized by a high-fat low-carbohydrate diet or focusing on carbohydrate intakes with a low glycemic index, high fiber content and whole grain consumption. Maintaining a reduced bodyweight, once achieved, also depends on glycemic load, with even further weight loss possible in prediabetic people with a low glycemic load maintenance diet. Maintenance of the achieved lower body weight is sustainable in normoglycemic persons using this approach. The results of the present study also suggest that overweight-obese individuals who do not yet show signs of diabetes and who aim to lose weight through a reduced fat intake, should consider additional intake of omega-3 LCPUFA in order to maintain sufficient dietary levels of these fatty acids. On the other hand, the study indicates that fiber intake may also need to be corrected in individuals losing weight on a low-glycemic index diet. A limitation of the current study is that the results are restricted to the population in which these were obtained, namely in Spain. The average omega-3 index is high, with one study reporting a value of 9.1% in the Spanish population. EPA+DHA intake clearly contributes to high RBC levels of EPA and DHA in this population, but only partly explains why the levels are relatively high, and other (unknown) environmental and dietary factors are likely contributing as well. The omega-3 index of the GLYNDIET participants was lower (~6.8%) and did not change during the 6-month weight loss intervention, except on the background of a low-fat diet, when the omega-3 LCPUFA status decreased. Further attention needs to be paid to the observation that ALA levels decreased in all tested weight loss protocols. The latter may relate to the fact that the Spanish population has a relatively high vegetable intake, and a lower energy intake overall may lead to reduced ALA-intake. A further limitation of this study relates to the estimation of fatty acid intake based on 3-day food records, which may significantly underestimate or skew the estimated intake of specific fatty acids that are infrequently consumed. Future studies that employ longer food records may possibly provide more reliable dietary intake estimates for specific fatty acids. It needs to be acknowledged that it is essentially impossible to alter any aspect of the diet without affecting something else, including the composition of fatty acids in the diet. Whereas olive oil was the main reported source of dietary fat in the low- and high-glycemic index diets, dairy was a main source of fat in the low fat diet group. Therefore, it remains a possibility that the changes in endogenous FA profile associated with the low fat diet could have been due to changes in the composition of ingested fatty acids, and which were not detected by diet records due to their lack of sensitivity.  Weight loss itself has been reported to be associated with changes in RBC fatty acid composition. In a small study, weight loss following subtotal gastrectomy markedly improved DHA tissue levels and omega-3 LCPUFA biosynthesis capacity of obese patients. A strong influence of body weight on omega-3 LCPUFA status following supplementation has also been recently uncovered in young people. The current results indicate that body weight reduction by itself was, however, not the main driver of changes in fatty acid composition, but rather that weight loss achieved by a low-fat diet may be associated with reductions in omega-3 LCPUFA, whereas a change in the type of carbohydrates during weight loss did not have such marked effects on LCPUFA status in non-diabetic overweight-obese adults. This study has provided a useful insight into the effect of different macronutrient compositions that can be provided during a weight loss schedule. Further studies that reduce confounding factors as much as possible may improve the already available approaches for achieving weight loss in individuals and different medical conditions, by minimizing the potential adverse effects on essential nutrient intake and their endogenous levels. Giardina S, Sala-Vila A, Hernández-Alonso P, Calvo C, Salas-Salvadó J, Bulló M. Carbohydrate quality and quantity affects the composition of the red blood cell fatty acid membrane in overweight and obese individuals. Clin. Nutr. 2017;Feb 11. [PubMed] Worth Noting Astrup A, Hjorth MF. Low-fat or low carb for weight loss? It depends on your glucose metabolism. EBioMedicine 2017. [PubMed] Christian LM, Young AS, Mitchell AM, Belury MA, Gracious BL, Arnold LE, Fristad MA. Body weight affects omega-3 polyunsaturated fatty acid (PUFA) accumulation in youth following supplementation in post-hoc analyses of a randomized controlled trial. PLoS One 2017;12(4):e0173087. [PubMed] Elizondo A, Araya J, Rodrigo R, Signorini C, Sgherri C, Comporti M, Poniachik J, Videla LA. Effects of weight loss on liver and erythrocyte polyunsaturated fatty acid pattern and oxidative stress status in obese patients with non-alcoholic fatty liver disease. Biol. Res. 2008;41(1):59-68. [PubMed] Fasting plasma glucose values: American Diabetes Association, Clinical Diabetes, 18, 2, 2000. [Link] Global Burden of Disease 2015 Obesity Collaborators, Health Effects of Overweight and Obesity in 195 Countries over 25 Years. N Engl J Med 2017;377(1):13-27. [PubMed] Gregg EW, Shaw JE. Global health effects of overweight and obesity. N. Engl. J. Med. 2017;377(1):80-81. [PubMed] Hjorth MF, Ritz C, Blaak EE, Saris WH, Langin D, Poulsen SK, Larsen TM, Sørensen TI, Zohar Y, Astrup A. Pretreatment fasting plasma glucose and insulin modify dietary weight loss success: results from 3 randomized clinical trials. Am. J. Clin. Nutr. 2017;July 5. [PubMed] Juanola-Falgarona M, Ibarrola-Jurado N, Salas-Salvadó J, Rabassa-Soler A, Bulló M. Design and methods of the GLYNDIET study; assessing the role of glycemic index on weight loss and metabolic risk markers. Nutr. Hosp. 2013;28(2):382-390. [PubMed] Juanola-Falgarona M, Salas-Salvado J, Ibarrola-Jurado N, Rabassa-Soler A, Diaz-Lopez A, Guasch-Ferre M, Hernandez-Alonso P, Balanza R, Bullo M. Effect of the glycemic index of the diet on weight loss, modulation of satiety, inflammation, and other metabolic risk factors: a randomized controlled trial. Am. J. Clin. Nutr. 2014;100(1):27-35. [PubMed] Mataix-Verdú J, Mañas-Almendros M, J. L-G, Martínez de Victoria-Muñoz E, Sänchez JJ, Borregón-Martínez A. Tabla de composición de alimentos españoles. Editorial Universidad de Granada. 2013; Campus Universitario de Cartuja.(5th ed.). [Link] Meinhold CL. Low-glycemic load diets: How does the evidence for prevention of disease measure up? J. Am. Diet Assoc. 2010;110(12):1818-1819. [PubMed] Meinhold CL, Dodd KW, Jiao L, Flood A, Shikany JM, Genkinger JM, Hayes RB, Stolzenberg-Solomon RZ. Available carbohydrates, glycemic load, and pancreatic cancer: is there a link? Am. J. Epidemiol. 2010;171(11):1174-1182. [PubMed] Prospective Studies Collaboration, Whitlock G, Lewington S, Sherliker P, Clarke R, Emberson J, Halsey J, Qizilbash N, Collins R, Peto R. Body-mass index and cause-specific mortality in 900 000 adults: collaborative analyses of 57 prospective studies. Lancet 2009;373(9669):1083-1096. [PubMed] Sala-Vila A, Harris WS, Cofán M, Pérez-Heras AM, Pintó X, Lamuela-Raventós RM, Covas MI, Estruch R, Ros E. Determinants of the omega-3 index in a Mediterranean population at increased risk for CHD. Br. J. Nutr. 2011;106(3):425-431. [PubMed]
Weight loss itself has been reported to be associated with changes in RBC fatty acid composition. In a small study, weight loss following subtotal gastrectomy markedly improved DHA tissue levels and omega-3 LCPUFA biosynthesis capacity of obese patients. A strong influence of body weight on omega-3 LCPUFA status following supplementation has also been recently uncovered in young people. The current results indicate that body weight reduction by itself was, however, not the main driver of changes in fatty acid composition, but rather that weight loss achieved by a low-fat diet may be associated with reductions in omega-3 LCPUFA, whereas a change in the type of carbohydrates during weight loss did not have such marked effects on LCPUFA status in non-diabetic overweight-obese adults. This study has provided a useful insight into the effect of different macronutrient compositions that can be provided during a weight loss schedule. Further studies that reduce confounding factors as much as possible may improve the already available approaches for achieving weight loss in individuals and different medical conditions, by minimizing the potential adverse effects on essential nutrient intake and their endogenous levels. Giardina S, Sala-Vila A, Hernández-Alonso P, Calvo C, Salas-Salvadó J, Bulló M. Carbohydrate quality and quantity affects the composition of the red blood cell fatty acid membrane in overweight and obese individuals. Clin. Nutr. 2017;Feb 11. [PubMed] Worth Noting Astrup A, Hjorth MF. Low-fat or low carb for weight loss? It depends on your glucose metabolism. EBioMedicine 2017. [PubMed] Christian LM, Young AS, Mitchell AM, Belury MA, Gracious BL, Arnold LE, Fristad MA. Body weight affects omega-3 polyunsaturated fatty acid (PUFA) accumulation in youth following supplementation in post-hoc analyses of a randomized controlled trial. PLoS One 2017;12(4):e0173087. [PubMed] Elizondo A, Araya J, Rodrigo R, Signorini C, Sgherri C, Comporti M, Poniachik J, Videla LA. Effects of weight loss on liver and erythrocyte polyunsaturated fatty acid pattern and oxidative stress status in obese patients with non-alcoholic fatty liver disease. Biol. Res. 2008;41(1):59-68. [PubMed] Fasting plasma glucose values: American Diabetes Association, Clinical Diabetes, 18, 2, 2000. [Link] Global Burden of Disease 2015 Obesity Collaborators, Health Effects of Overweight and Obesity in 195 Countries over 25 Years. N Engl J Med 2017;377(1):13-27. [PubMed] Gregg EW, Shaw JE. Global health effects of overweight and obesity. N. Engl. J. Med. 2017;377(1):80-81. [PubMed] Hjorth MF, Ritz C, Blaak EE, Saris WH, Langin D, Poulsen SK, Larsen TM, Sørensen TI, Zohar Y, Astrup A. Pretreatment fasting plasma glucose and insulin modify dietary weight loss success: results from 3 randomized clinical trials. Am. J. Clin. Nutr. 2017;July 5. [PubMed] Juanola-Falgarona M, Ibarrola-Jurado N, Salas-Salvadó J, Rabassa-Soler A, Bulló M. Design and methods of the GLYNDIET study; assessing the role of glycemic index on weight loss and metabolic risk markers. Nutr. Hosp. 2013;28(2):382-390. [PubMed] Juanola-Falgarona M, Salas-Salvado J, Ibarrola-Jurado N, Rabassa-Soler A, Diaz-Lopez A, Guasch-Ferre M, Hernandez-Alonso P, Balanza R, Bullo M. Effect of the glycemic index of the diet on weight loss, modulation of satiety, inflammation, and other metabolic risk factors: a randomized controlled trial. Am. J. Clin. Nutr. 2014;100(1):27-35. [PubMed] Mataix-Verdú J, Mañas-Almendros M, J. L-G, Martínez de Victoria-Muñoz E, Sänchez JJ, Borregón-Martínez A. Tabla de composición de alimentos españoles. Editorial Universidad de Granada. 2013; Campus Universitario de Cartuja.(5th ed.). [Link] Meinhold CL. Low-glycemic load diets: How does the evidence for prevention of disease measure up? J. Am. Diet Assoc. 2010;110(12):1818-1819. [PubMed] Meinhold CL, Dodd KW, Jiao L, Flood A, Shikany JM, Genkinger JM, Hayes RB, Stolzenberg-Solomon RZ. Available carbohydrates, glycemic load, and pancreatic cancer: is there a link? Am. J. Epidemiol. 2010;171(11):1174-1182. [PubMed] Prospective Studies Collaboration, Whitlock G, Lewington S, Sherliker P, Clarke R, Emberson J, Halsey J, Qizilbash N, Collins R, Peto R. Body-mass index and cause-specific mortality in 900 000 adults: collaborative analyses of 57 prospective studies. Lancet 2009;373(9669):1083-1096. [PubMed] Sala-Vila A, Harris WS, Cofán M, Pérez-Heras AM, Pintó X, Lamuela-Raventós RM, Covas MI, Estruch R, Ros E. Determinants of the omega-3 index in a Mediterranean population at increased risk for CHD. Br. J. Nutr. 2011;106(3):425-431. [PubMed]

